Science- First Building - Ground Floor
Responsible Prof. Francesco Spinozzi
Research activities
The scientific activities carried on in the Molecular Biophysics Laboratory concern the characterization of the nanometric structures formed by the main biological molecules in aqueous solutions.
The experimental techniques mostly used by the Molecular Biophysics Laboratory, especially through innovative approaches for data analysis, are small-angle scattering (SAS) of X-rays (SAXS) and neutrons (SANS) and X-ray diffraction (XRD). These measurements are carried out at the most advanced instruments present in national and international Large Scale Facilities (LSF): synchrotrons (SAXS) and neutron sources (SANS).
Other techniques employed are: dynamic light scattering (DLS), differential scanning calorimetry (DSC), fluorescence spectroscopy, differential scanning calorimetry (DSC), Fourier transform infrared spectroscopy (FTIR), Raman spectroscopy, atomic force microscopy (AFM), phase-contrast optical microscopy and gravimetry with quartz crystal microbalance.


Main research lines
The main research lines of the Molecular Biophysics Laboratory include:
-
self-assembly mechanisms of guanosines and their derivatives;
-
structure and stability of lipid phases;
-
structure, stability, and aggregation processes of proteins in solution.
Guanosine self-assembly
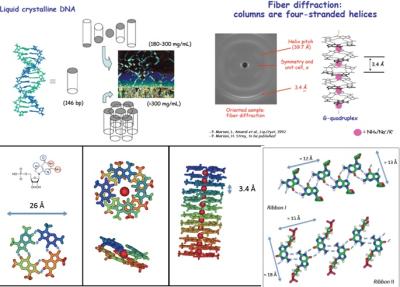
In this research line, self-assembly processes of guanosine and of its derivatives (lipophilic, too), which form macroaggregates constituted by four-stranded helical filaments (quadruplexes), are investigated. The aggregation of short-chain of double-stranded DNA and single-stranded DNA, which are partially complementary and therefore capable to form nanostars structures with different valence, are object of our research, too. These systems are the subject of an intense theoretical and experimental work, which concerns:
-
the structural properties of four-stranded helices;
-
the thermodynamic aspects that determine length and stability;
-
the mechanism of hierarchical self-assembly in dilute solutions;
-
the switching between structural nanoribbons and nanowires observed in organic solvents in the presence and absence of counterions;
-
the transmission mechanism of the long-range order;
-
the formation of lyotropic liquid-crystalline phases;
-
the propagation of chirality;
-
the condensation promoted by counterions in aqueous solution;
-
the separation of phase in osmolytes and the formation of hydrogels.
It is worth to note that, from a biological point of view, the formation of quadruplexes can be correlated with some biophysical properties of chromatin, with the mechanism of action of telomerase, with some anticancer drugs and even with the source of the prebiotic DNA molecule.
Guanosine hydrogel
This line of research is focused on the study of a system of G-quadruplexes formed by mixtures of guanosine (GMP) and guanine (Gua), a molecule with the same nitrogenous base as GMP. The helical structures that self-assemble through the mechanism of π-stacking interactions interact on average more attractively than the structures formed by samples containing only GMP, since the Gua molecules are electrically neutral. The presence of stable helix-helix interactions thus leads to the formation of a physical hydrogel, with a complex supramolecular architecture.
Techniques such as atomic force microscopy (AFM), scanning electron microscopy (SEM), UV-Vis spectroscopy, SAXS and SANS are very useful for the structural characterization of this hydrogel. For example, it is possible to study how the physical-chemical properties of the self-assembled system change as a function of the stoichiometric ratio GMP:Gua in the mixture.
Another important part of the research concerns the study of biotechnological applications in which the G-hydrogel could be implicated. It is known that similar systems are used in the tissue engineering for skin regeneration, in the pharmaceutical sector as drug carriers and so on.
Currently, two possible applications are being studied in the Laboratory of Molecular Biophysics:
-
study of the diffusion of fluorescent molecules incorporated within the G-hydrogel to investigate its dynamic properties;
-
use of G-hydrogel as a carrier of protein micro-crystals directly on the X-ray beam, in order to derive theirs structure (collaboration with the Dynamop group at the Institute de Biologie Structurale in Grenoble).

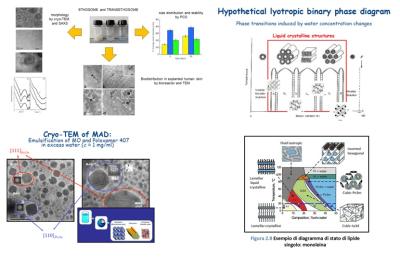
Structure and stability of lipid phases
The aspects of lipid systems that we investigate concern:
-
the structural characterization of the phases and the lyotropic polymorphism as a function of different experimental parameters (such as, composition, temperature, mechanical pressure, presence of counterions, pH, organic solvents);
-
the mechanisms of growth and transformation of macromolecular aggregates;
-
the stability of the different phases and of the aggregates themselves (based on analysis of different energy and molecular contributions);
-
the hydration properties connected with the self-assembly and to the polymorphic behavior (with particular reference to non-lamellar lipid phases);
-
defects in model membranes due to the spontaneous formation of water pores of to the presence of membrane proteins investigated by analysing SAXS data with innovative models based on the possible presence of horizontal correlations among these defects.
Recently, this research line has increasingly focused on the problem of drug delivery, by studying the structural properties and the thermal stability of lipid nanoparticles exploited as biocompatible and stable drug carriers.
The research activities concern the solid-lipid nanoparticles, the cubosomes (obtained by sonication after adding suitable surfactants from cubic phases of monoglycerides) and hexosomes (also obtained by sonication from hexagonal phases lipid in the presence of suitable surfactants).
Giant unilamellar vesicles
Another particular system investigated in our laboratory belongs to Giant Unilamellar Vesicles (GUVs) with size comparable to that of the cell (diameter 1-100 μm), which can be observed with the optical microscope. GUVs constitute an extremely useful and versatile tool for the in vitro study of the properties of lipid membranes and their interaction with biological macromolecules, since by varying the lipid composition and inserting specific components (such as cholesterol and proteins) the main features of plasma membranes can be easily mimed.
At the Molecular Biophysics Laboratory a system for the production of GUVs has been developed, based on the electroporation process in a flow chamber. An optical system (camera and digital camera) for recording images obtained with the polarizing microscope is available.
Recently, the research activity concerns the thermal and osmotic stability of the GUVs and their behaviour by varying chemical-physical conditions, such as:
-
lipid composition or temperature
-
interactions with biosurfactants (mono- and di-rhamnolipids)
-
interactions with amyloid protofibrils or with amyloid fibrils.

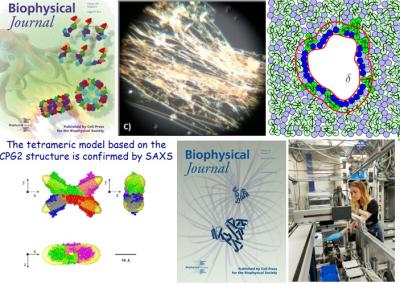
Structure, stability, aggregation and interaction of proteins in solution
This research line concerns the investigation of the structural properties of proteins in solution (including model proteins, proteins not yet crystallized and protein in non-native conditions) by means of small-angle X-rays and neutron scattering (SAXS and SANS, SAS in general) techniques. We study the the following properties:
-
shape reconstruction of proteins;
-
interaction between proteins in solution;
-
structural properties of proteins subjected to mechanical pressure;
-
aggregation properties of proteins in solution;
-
proteins in mixed solvents;
-
prediction of protein structures by SAS and new bioinformatics approaches;
-
amyloid formation processes;
-
proteins trapped on polymeric layers;
-
aggregation of proteins consisting of domains with known tertiary structure and flexible connections with known primary structure;
-
variational Bayesian methods for studying intrinsically disordered proteins.
In such studies, innovative strategies for data analysis were developed, which allow obtaining more detailed structural and thermodynamic information than those provided by traditional approaches.
The developed methods, mainly based on global fitting approaches, have been collected in a suite of shareware software: GENFIT, SASMOL (downloadable from the website https://sites.google.com/site/genfitweb/), QUAFIT, VBWSAS, MPOLE and GRAFIT (available upon request at e-mail).
In the last decade we extensively studied proteins linked to neurodegenerative diseases, like Alzheimer and Parkinson, which we studied in the presence of biological (chaperonins) and chemical (curcumin-like molecules) agents for the design of new therapeutic approaches to these pathologies. We are currently studying the inhibitory capacity in the pathological aggregation of proteins by products deriving from agricultural waste, in a circular economy perspective.
We have developed new methods to investigate the properties of intrinsically disordered proteins (IDPs) by combining SAXS or SANS measurements with conformational ensembles determined by Molecular Dynamics or Monte Carlo techniques.
Nowadays, we are involved in several collaboration involving proteins linked to SARS-CoV-2 and to rare diseases.

Application of FTIR and Raman spectroscopies to physical, chemical, and biological systems
FTIR and Raman techniques are vibrational spectroscopies that allow the study of the chemical composition of a sample thanks to the vibrational modes specific of each chemical bond. These techniques, also coupled with optical microscopes – FTIR and Raman Microspectroscopies – let obtain the fingerprint of the analyzed sample in an objective and rapid way. The fields of application of these techniques are countless, and among those we are focused on there are:
-
evaluation of the physico-chemical properties of novel bioactive systems/compounds, for drug design and vehiculation;
-
study of the drug/cell interaction: detection of cellular biochemical changes in response to drug action, in-vitro drug tracking and sub-cellular compartments analysis;
-
development of spectral pathology methods, based on FTIR and Raman Imaging for improving routine pathology procedures: detection of chemical changes in tissue samples that precede or accompany morphological modifications in biological systems;
-
physico-chemical characterization of the structural organization and features of collagen in human diseases; analysis of the involvement of collagen in the tumor microenvironment."
Main current research projects
We are involved in 2 PNRR Research Projects.
The first one, VITALITY, concerns the ecosystem for innovation, digitalisation, and sustainability for the economy of central Italy and focuses on nano-bio autoassembly materials.
The second one, National Biodiversity Future Center, focuses on the development of sensors able to reveal very low amounts on new pollution agents.
Laboratory equipment
Centrifuge Hermle Z200A for falcon - complete with rotor
Microcentrifuge ALC 4214
Mettler AE163 analytical balance
BioRAD electrophoresis systems
Bransonic ultrasound bowl
Zeiss stereo microscope
SpeedVAC for concentration/evaporation
USF PureLab Distilled Water Manufacturer - Steroglass
Generalcontrol refrigerated incubator
Image plate scanner, DENOPTIX
Leitz Ortolux polarized light microscope with temperature control and digital camera
Leica DMLS phase contrast microscope
Philips DY673 generator with monochromator and Guiner chamber with Image Plate
Ital-Structures C3K5 generator with monochromator and Guinier chamber with Image Plate
Nikon D5100 digital camera
Edwards freeze dryer
Electroporation system and cells with electrodes
Staff
|
Paolo Mariani |
 |
|
Prof. Francesco Spinozzi |
 |
|
Dr. Maria Grazia Ortore |
 |
| Dr. Valentina Notarstefano |  |
| Dr. Paolo Moretti |  |
| Dr. Alessia Pepe |  |
| Dr. Adnan Khan |  |
| Dr. Eleonora Mari |  |
| Dr. Matteo Vercelli |  |
| Dr. Jessica Roque |  |