|
Organic chemistry and Chemical biology with non natural molecules
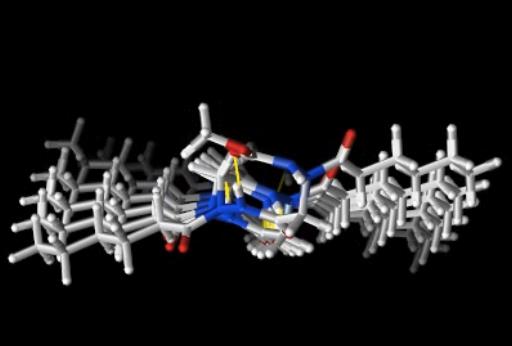
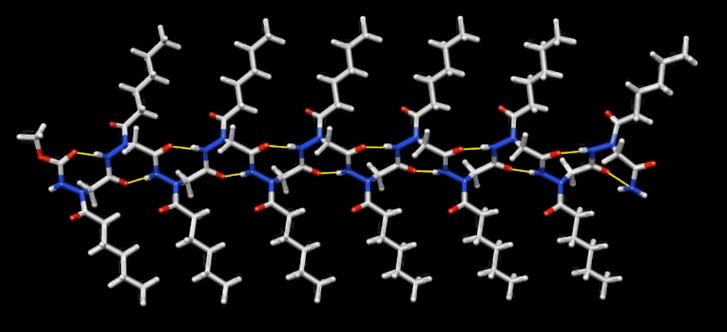
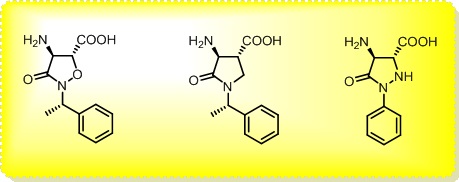
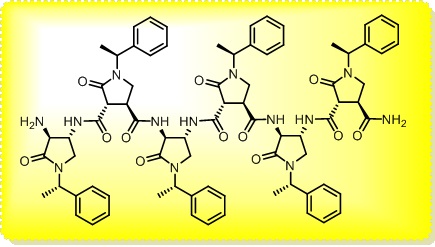
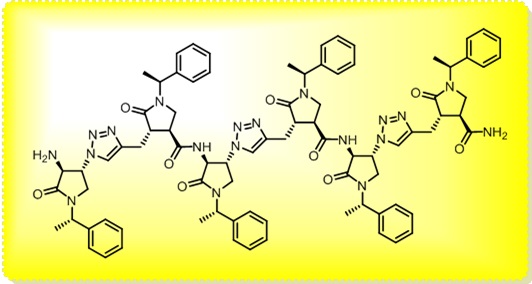
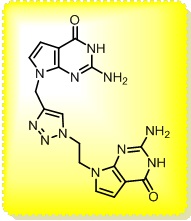
The main interest of the research group is directed towards the synthesis of small, polyfunctionalized organic molecules, in particular β-amino acids and nitrogen containing heterocycles, in order to obtain novel compounds with definite biological activity. In particular, we envisaged the synthesis of peptidomimetics containing analogues of natural amino acids, in order to identify new bioactive structures leading selectively to cytotoxic drugs, enzyme inhibitors or diagnostic tools for identification of tumor cells. The biological activity of the synthesized products is evaluated in collaboration with research groups having a recognized expertise in their specific fields. In addition, for some years we were engaged in the synthesis of organic compounds of medium size (500 to 3000 amu approximately) pseudopeptidic structure, where natural amino acids are completely or partially replaced by synthetic structures chosen to impose a local conformational restriction which favors the formation of ordered secondary structure. These compounds, which are defined foldamers, can have many applications, both as pharmacologically active compounds (for an example, they are able to inhibit protein-protein interactions) and as new supramolecular materials with interesting properties. Our group currently synthesizes, purifies and analyzes these compounds, which are useful for a lot of applications in chemical biology. At present we are developing new monomers and oligomers containing conformational constraints sterically ordered that form helices of various sizes. (click on images to enlarge)
|
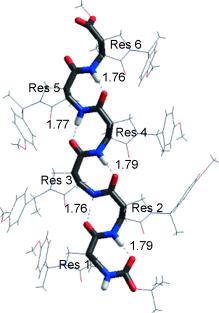
Helix-8
|